The Sunflower Crop Survey is led by the National Sunflower Association and carried out by a network of volunteer from universities, government, producers and industry, including Manitoba Crop Alliance (MCA). Participating regions include Manitoba, North Dakota, South Dakota, Minnesota, Nebraska, Kansas, and Colorado, where data is collected on a number of variables and compiled to document sunflower growing conditions, pest challenges and yield. The survey is conducted on alternate years, with fall 2025 being the most recent.
Manitoba and U.S. Survey Overview:
191 Sunflower fields were samples across all participating regions. The following management practices were observed:
- Sunflower type: 7% confection; 93% oilseed
- Water Management: 2% irrigated; 98% dryland
- Tillage: 24% conventional; 21% minimum; 55% no-till
The survey has a large focus on pest pressures. The cumulative pests that were monitored across all locations were as follows:
- 34% of fields had blackbird damage
- 11% of fields had seed maggot damage
- 8% of fields had bud moth damage
- 8% of fields had sunflower midge damage
- 14% of fields had long-horned beetle damage. It is important to note that long-horned beetle has not been found in Manitoba during this survey, or in anecdotal scouting events, however there are fields sampled in this survey that are extremely close to the MB-ND border that had long-horned beetle (dectes) damage in both 2025 and 2023.
Manitoba Overview:
12 sunflower fields in 10 municipalities were sampled in Manitoba from the RM of Brokenhead to the RM of Two Borders:
- Sunflower type: 100% oilseed
- Water Management: 100% dryland
- Tillage: 25% conventional; 67% minimum; 8% no-till
- Row Spacing: 50% had 20” or narrower row spacing, 50% had 22” to 30” row spacing
Each field was surveyed in two locations and a small sample area of two rows by 25 feet was used to gather data in each of the two locations. Pests were recorded, full plants were assessed, and seed samples were taken.
The highest yield in a sampled area was 2,792 lb/ac.
The lowest yield in a sampled area was 983 lb/ac.
The average yield among all 12 fields sampled was 2,000 lb/ac.
Yield-Limiting Factors
- Seven of the 12 fields were limited mainly by disease, in general.
- One of the 12 fields was limited mainly by bird presence and feeding.
- One of the 12 fields was limited mainly by row spacing, as it was a solid-seeded field. This was hard to measure yield on such a small scale with fewer plants per foot of row.
- Three of the 12 fields were limited mainly by weed pressure, with one of them being specifically limited by kochia infestation. In the 12 fields, weed pressure was generally quite low and not at all a concern in 75% of the fields.
- Secondary limitations in the 12 fields sampled included wildlife (i.e., elk), lodging, insect, sclerotinia and birds.
Disease Presence and Severity
Sclerotinia:
- Sclerotinia basal stem rot was minor in all fields sampled. Half of the fields had no basal wilt present. One field had up to eight plants with basal wilt symptoms, which would be estimated at about 8% of the sample size in that field.
- Sclerotinia mid-stalk rot presence was similar to basal stem rot. Eight fields had zero or just one plant infected; one field had six infections and another had seven infections; two fields had 10 or 12 plants infected, respectively.
- Sclerotinia head rot were higher, in general. Half of the fields had four or less infected heads; three fields had 5-7 infected heads, one field had 10 infected heads, one field had 12 infected heads and one field had 22 infected heads. This last field did report the lowest yield as well, unsurprisingly.
Downy Mildew:
- Low incidence among all fields, except one that had four affected plants
Phomopsis:
- Six of the 12 fields had almost negligible one or zero plants with Phomopsis infections.
- Five of the 12 fields had from six to 15 stalks infected with Phomopsis.
- One field had 40 plants with Phomopsis infections, which was roughly 80% incidence in that location.
Phoma:
- Incidence was much higher and present in relatively high numbers in each of the 12 fields, except for two that had zero incidence.
- The three highest fields of incidence had 32, 42 and 46 plants with Phoma stem infections.
Rhizopus:
Rhizopus has not been a disease of concern in Manitoba, simply because it can largely go undetected. The last time the sunflower survey was conducted, in 2023, three of eight fields had Rhizopus in the sample set.
Rhizopus can be identified on a sunflower head by the presence of gray, fuzzy mycelium, usually viewed on the face and between developed seeds. The disease prefers warm, humid environments and most often originates via wounds on the back of the sunflower head. Infections do affect yield by limiting seed fill and potentially causing head drop in severe cases.
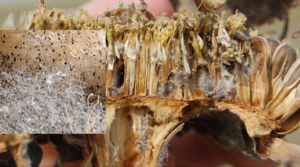
Rhizopus on sunflower head. Photo courtesy of North Dakota State University.
Sunflower Rust:
Sunflower rust was very minimal in 2025 and was found in four of the 12 fields at very minor severity in September. The highest severity was one field that exhibited 0.75% of leaf area on the top four leaves being affected by rust pustules.
Verticillium Wilt:
Verticillium wilt is also uncommon in sunflower fields in Manitoba, though it was found in the 2025 survey. Symptoms can include leaf mottle, or interveinal chlorosis, and a greenish discolouration on the stem, where further inspection is required. By splitting the stem at the base of the plant, discoloured vascular tissue is visible around the pith.
- Three fields were found to have Verticillium wilt with five, six and 12 plants infected, respectively.
Insect Presence and Severity
Sunflower Midge:
- Seven of the 12 fields had Sunflower Midge damage.
- The field with the greatest damage had seven affected heads.
Sunflower Seed Maggot:
- Three of 12 fields had Sunflower Seed Maggot damage.
- Each of the three fields had one plant affected.
Sunflower Bud Moth:
- Nine of 12 fields had Sunflower Bud Moth damage, specifically on the sunflower head.
- The field with the greatest damage had 14 affected heads, followed by a field that had 10 affected heads.
- The remaining seven fields had minor damage noted.
Blackbird Presence and Severity
Blackbird feeding was noted in five of the 12 fields sampled. Assessment is based on the area of the sunflower head with missing seed that has recognizably been fed on by birds. Seed is usually completely missing and sunflower seed shells may be found on or near the plant in question.
All four fields with damage noted were quite minor in the sampled areas, the greatest having about 6.35% of the total head area missing seed.
Other Yield Factors
Actual plant populations of sampled fields ranged from 12,200 to 24,400 plants per acre. Strangely enough, the lowest populated field also had the smallest head size, averaging about 5.75” in diameter. The field with largest head size overall was 8.45” diameter. Generally, head size was smaller in the sampled fields than an average year would see, but this may have been a result of dry growing conditions during head development.
Seed size was reported to be good to excellent and seed fill ranged from 70 – 99% across the 12 fields. Centre seed set was reasonable, but there were some fields that did have up to two inches of the head centre undeveloped, which drastically affects yield.
MCA would like to thank all 12 participants of the Sunflower Survey for allowing us to use your fields for this project. Also, thank you to Manitoba Agriculture oilseeds specialist Sonia Wilson and crop production extension specialist Callum Morrison for your help surveying several fields. Finally, thank you to Dr. Ahmed Abdelmagid, research scientist and oilseed pathologist at AAFC Morden, for surveying several fields and collecting various samples of sunflower diseases to understand the scope of disease presence in Manitoba.